
After the acid exposure, the alloy is exposed to oxygen in order to trigger the formation of the protective anti-corrosive barrier. The passivation process starts by applying acid to the stainless steel compound in order to remove the free iron molecules from the alloy’s surface, while chromium remains unaffected. The strong anti-corrosive properties of chromium come forth when this chemical element is exposed to oxygen molecules as a part of the stainless steel alloy. Also, molybdenum, silicon, manganese, aluminum, and carbon may be added to this alloy. The combination of chromium, iron, and nickel as primary elements results in the production of stainless steel. When exposed to high temperatures, the pure elemental form of chromium burns and forms the green chromium oxide compound. 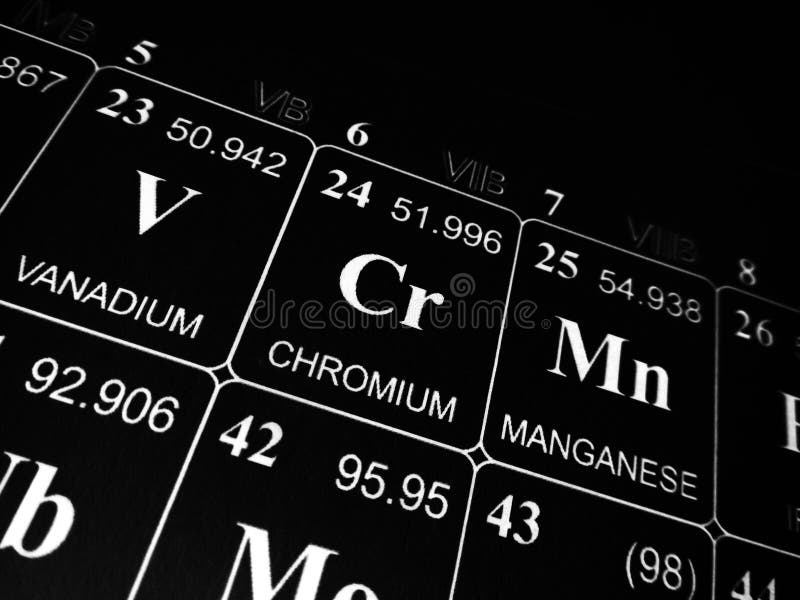
Highly reactive with the other chemical elements, chromium displays an electronegativity of 1.6 according to Pauling, whereas its atomic radius according to van der Waals is 0.127 nm. Also, it’s the only element that does not display any magnetic properties in its solid form. After the elements carbon and boron, chromium is the third hardest chemical element in the periodic table. Apart from being resistant to heat, wear, and corrosion, chromium is also classified as a refractory metal, along with the elements molybdenum (Mo) and tungsten (W). This lustrous, brittle, and corrosion-resistant hard metal can be polished to a highly reflective surface. It reaches its boiling point at 2672 ☌, while the melting point is achieved at 1907 ☌. 
The symbol in the periodic table of elementsĪ lustrous silvery-gray metal with a bluish hueĬhromium is a transition metal assigned with the periodic table symbol Cr, atomic number 24, has an atomic mass of 51.996 g.mol -1, and electron configuration 3d 5 4s 1. Chemical and Physical Properties of Chromium Property
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
